 
The negatively charged plums were surrounded by positively charged pudding.Ī few years later, Ernest Rutherford , one of Thomson’s students, did some tests on Thomson’s plum pudding model. This became known as the ‘plum pudding’ model of the atom. So, he determined that they were surrounded by positively-charged material. 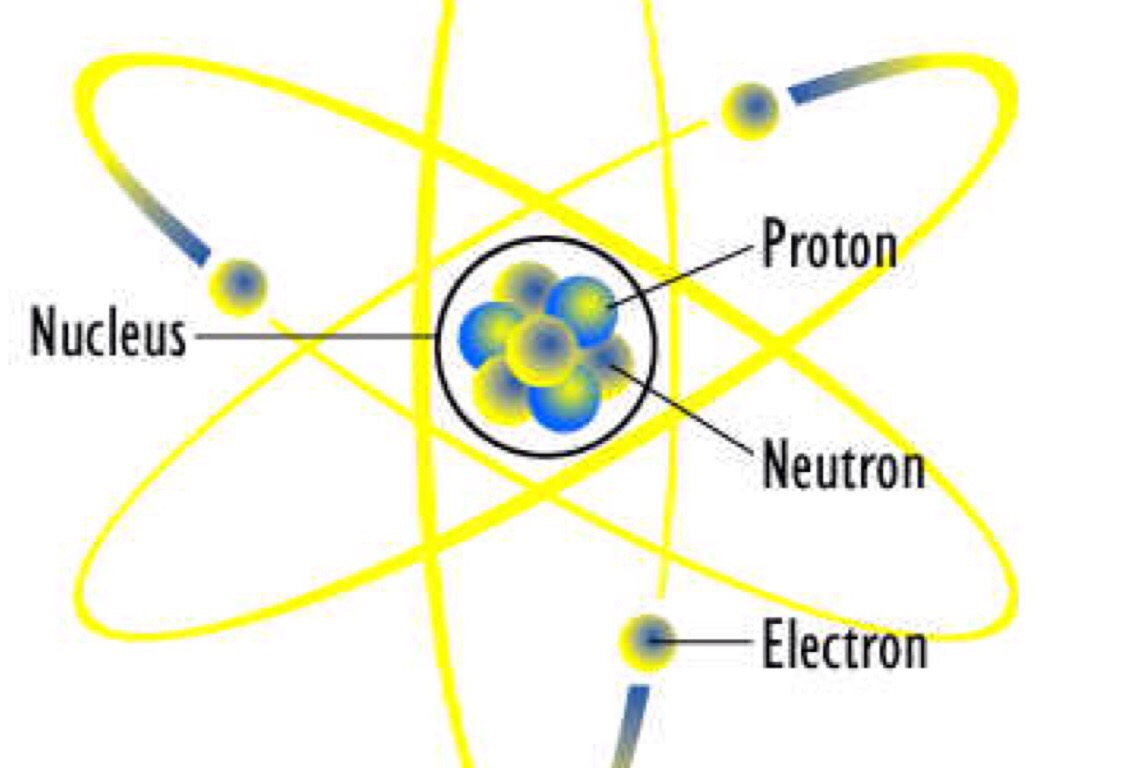
Thomson went one step further and determined that these negatively charged electrons needed something positive to balance them out. From this he decided that these particles must have come from somewhere within the atom and that Dalton was incorrect in stating that atoms cannot be divided into smaller pieces. He performed many experiments and found that the mass of one of these particles was almost 2,000 times lighter than a hydrogen atom. Thomson used the cathode ray tube with a magnet and discovered that the green beam it produced was made up of negatively charged material. You’ve probably seen a cathode ray tube without even knowing it! They are the bulky electronic part of old television sets. Thomson used what was called a cathode ray tube, or an electron gun. 
Now, let’s fast-forward to the late 1800s when J.J. His model was created solely on experiments that were macroscopic, or seen with the unaided eye. The really awesome thing about Dalton’s model of the atom is that he came up with it without ever seeing the atom! He had no concept of protons, neutrons or electrons. He also stated that all atoms of the same element will be exactly the same and that atoms of different elements can combine to form compounds. Dalton went on to say that matter is made up of tiny particles, called atoms, that cannot be divided into smaller pieces and cannot be destroyed. It wasn’t until around 2,000 years later, in the early 1800s, when John Dalton came along and disproved Aristotle. So, who was right? Aristotle was very convincing and did many experiments using the scientific method, so more people believed him. He thought that matter could only be divided until you got to the smallest particle (which he called the atom, coming from the Greek word atomos, meaning indivisible). The Greek philosopher Aristotle believed that matter could be divided infinitely without changing its properties. This image is a popular icon of the atom, but it only vaguely represents our current model of what the atom looks like.įirst, we are going to travel back a little over 2,000 years ago to the times of Aristotle and Democritus. What does it look like? Most likely it will resemble something like this: a fairly large nucleus surrounded by orbiting electrons whizzing around the nucleus. In this lesson, we are going to travel back in time and discuss some of the major discoveries in the history of the atom. Imagine firing a bullet at a piece of tissue paper and having it bounce back at you! You would probably be just as surprised as Rutherford when he discovered the nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
